Elements, Compounds, and Mixtures
By Zara Arales
Elements
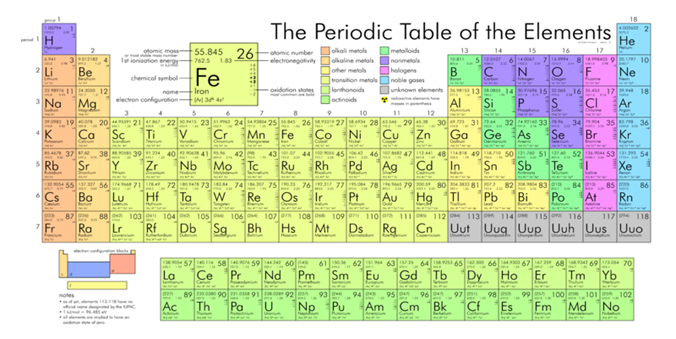
Elements are the simplest form of matter that has a unique set of properties. Element is a substance that cannot be broken down by chemical means and defined by the number of proton they possess. Elements are divided into metals, metalloids and non-metals. Elements can be found on the periodic table. Elements may be thought of as the basic chemical building blocks of matter. Depending on how much evidence you require to prove a new element has been created, there are 120 known elements.
Diatomic molecule is a molecule containing 2 atoms of the same element And the seven diatomic elements are:
Hydrogen (H2)
Nitrogen (N2)
Oxygen (O2)
Fluorine (F2)
Chlorine (Cl2)
Iodine (I2)
Bromine (Br2)
Properties of an element
Properties of an element are sometimes classed as either chemical or physical. Chemical properties are usually observed in the course of a chemical reaction, while physical properties are observed by examining a sample of the pure element. The chemical properties of an element are due to the distribution of electrons around the atom's nucleus, particularly the outer, or valence, electrons; it is these electrons that are involved in chemical reactions. A chemical reaction does not affect the atomic nucleus; the atomic number therefore remains unchanged in a chemical reaction.
Compound
A compound is a pure chemical substance consisting of two or more different chemical elements that can be separated into simpler substances by chemical reactions. Chemical compounds have a unique and defined chemical structure; they consist of a fixed ratio of atoms that are held together in a defined spatial arrangement by chemical bond . Chemical compounds can be molecular compounds held together by covalent bonds , salts held together by ionic bonds, intermetallic compounds held together by metallic bonds, or complexes held together by coordinate covalent bonds. Pure chemical elements are not considered chemical compounds, even if they consist of molecules that contain only multiple atoms of a single element which are called diatomic molecules or polyatomic molecules.
Properties of a Compound
A compound has unique properties that are distinct from the properties of its elemental constituents. One familiar chemical compound is water, a liquid that is nonflammable and does not support combustion. It is composed of two elements: hydrogen, an extremely flammable gas, and oxygen, a gas that supports combustion. A compound differs from a mixture in that the components of a mixture retain their own properties and may be present in many different proportions. The components of a mixture are not chemically combined; they can be separated by physical means. A mixture of hydrogen and oxygen gases is still a gas and can be separated by physical methods. If the mixture is ignited, however, the two gases undergo a rapid chemical combination to form water. Although the hydrogen and oxygen can occur in any proportion in a mixture of gases, they are always combined in the exact proportion of two atoms of hydrogen to one atom of oxygen when combined in the compound water. Another familiar compound is sodium chloride (common salt). It is composed of the silvery metal sodium and the greenish poisonous gas chlorine combined in the proportion of one atom of sodium to one atom of chlorine
Mixture
Physical combinations of 2 or more substance in which amount may greatly vary. Components retain their own composition and properties. May be separated into pure substance by physical methods. A mixture is a material system made up of two or more different substances which are mixed but are not combined chemically. A mixture refers to the physical combination of two or more substances on which the identities are retained and are mixed in the form of alloys, solutions suspensions and colloids
Mixtures are the product of a mechanical blending or mixing of chemical substances like elements and compounds, without chemical bonding or other chemical change, so that each ingredient substance retains its own chemical properties and makeup. Despite that there are no chemical changes to its constituents, the physical properties of a mixture, such as its melting point, may differ from those of the components. Some mixtures can be separated into their components by physical (mechanical o thermal) means. Zoetrope’s can be considered as a kind of mixture which usually pose considerable difficulties regarding the separation processes required to obtain their constituents (physical or chemical processes or, even a blend of them).
Properties of a Mixture
1) There is no fixed composition in mixtures.
2) Mixtures do not have a fixed boiling point.
3) Mixtures do not have a fixed boiling point.
Exercises:
Tell whether if it is an element, a compound or a mixture
1. Nitrogen
2. chlorine
3. sugar and salt
4. Silicon
5. carbon dioxide
6. Hydrogen
7. sodium chloride
8. Oxygen
9. gold
10. acetic acid
11. Magnesium
12. calcium
13. sand and water
14. copper
15. salt and water
By Zara Arales
Elements
Elements are the simplest form of matter that has a unique set of properties. Element is a substance that cannot be broken down by chemical means and defined by the number of proton they possess. Elements are divided into metals, metalloids and non-metals. Elements can be found on the periodic table. Elements may be thought of as the basic chemical building blocks of matter. Depending on how much evidence you require to prove a new element has been created, there are 120 known elements.
Diatomic molecule is a molecule containing 2 atoms of the same element And the seven diatomic elements are:
Hydrogen (H2)
Nitrogen (N2)
Oxygen (O2)
Fluorine (F2)
Chlorine (Cl2)
Iodine (I2)
Bromine (Br2)
Properties of an element
Properties of an element are sometimes classed as either chemical or physical. Chemical properties are usually observed in the course of a chemical reaction, while physical properties are observed by examining a sample of the pure element. The chemical properties of an element are due to the distribution of electrons around the atom's nucleus, particularly the outer, or valence, electrons; it is these electrons that are involved in chemical reactions. A chemical reaction does not affect the atomic nucleus; the atomic number therefore remains unchanged in a chemical reaction.
Compound
A compound is a pure chemical substance consisting of two or more different chemical elements that can be separated into simpler substances by chemical reactions. Chemical compounds have a unique and defined chemical structure; they consist of a fixed ratio of atoms that are held together in a defined spatial arrangement by chemical bond . Chemical compounds can be molecular compounds held together by covalent bonds , salts held together by ionic bonds, intermetallic compounds held together by metallic bonds, or complexes held together by coordinate covalent bonds. Pure chemical elements are not considered chemical compounds, even if they consist of molecules that contain only multiple atoms of a single element which are called diatomic molecules or polyatomic molecules.
Properties of a Compound
A compound has unique properties that are distinct from the properties of its elemental constituents. One familiar chemical compound is water, a liquid that is nonflammable and does not support combustion. It is composed of two elements: hydrogen, an extremely flammable gas, and oxygen, a gas that supports combustion. A compound differs from a mixture in that the components of a mixture retain their own properties and may be present in many different proportions. The components of a mixture are not chemically combined; they can be separated by physical means. A mixture of hydrogen and oxygen gases is still a gas and can be separated by physical methods. If the mixture is ignited, however, the two gases undergo a rapid chemical combination to form water. Although the hydrogen and oxygen can occur in any proportion in a mixture of gases, they are always combined in the exact proportion of two atoms of hydrogen to one atom of oxygen when combined in the compound water. Another familiar compound is sodium chloride (common salt). It is composed of the silvery metal sodium and the greenish poisonous gas chlorine combined in the proportion of one atom of sodium to one atom of chlorine
Mixture
Physical combinations of 2 or more substance in which amount may greatly vary. Components retain their own composition and properties. May be separated into pure substance by physical methods. A mixture is a material system made up of two or more different substances which are mixed but are not combined chemically. A mixture refers to the physical combination of two or more substances on which the identities are retained and are mixed in the form of alloys, solutions suspensions and colloids
Mixtures are the product of a mechanical blending or mixing of chemical substances like elements and compounds, without chemical bonding or other chemical change, so that each ingredient substance retains its own chemical properties and makeup. Despite that there are no chemical changes to its constituents, the physical properties of a mixture, such as its melting point, may differ from those of the components. Some mixtures can be separated into their components by physical (mechanical o thermal) means. Zoetrope’s can be considered as a kind of mixture which usually pose considerable difficulties regarding the separation processes required to obtain their constituents (physical or chemical processes or, even a blend of them).
Properties of a Mixture
1) There is no fixed composition in mixtures.
2) Mixtures do not have a fixed boiling point.
3) Mixtures do not have a fixed boiling point.
Exercises:
Tell whether if it is an element, a compound or a mixture
1. Nitrogen
2. chlorine
3. sugar and salt
4. Silicon
5. carbon dioxide
6. Hydrogen
7. sodium chloride
8. Oxygen
9. gold
10. acetic acid
11. Magnesium
12. calcium
13. sand and water
14. copper
15. salt and water