Matter and its properties
By Jason Angeles
Matter
Matter is anything that occupies space and has mass. Basically, there are Three phases of matter:
Solid - their molecules are more compact and have strong inter-molecular attraction and have less kinetic energy meaning there movement is restricted and can only vibrate, like rocks, metal and ice.
Liquid - liquids have indefinite shape, their molecules are less compact meaning that they can move more freely than solids, and have low kinetic energy. Examples are water, coca cola, and alcohol.
Gas – Gas is the 3rd phase of matter. Their molecules are scattered, meaning they can move more freely and have high kinetic energy. Examples are Oxygen, Carbon dioxide, and helium.
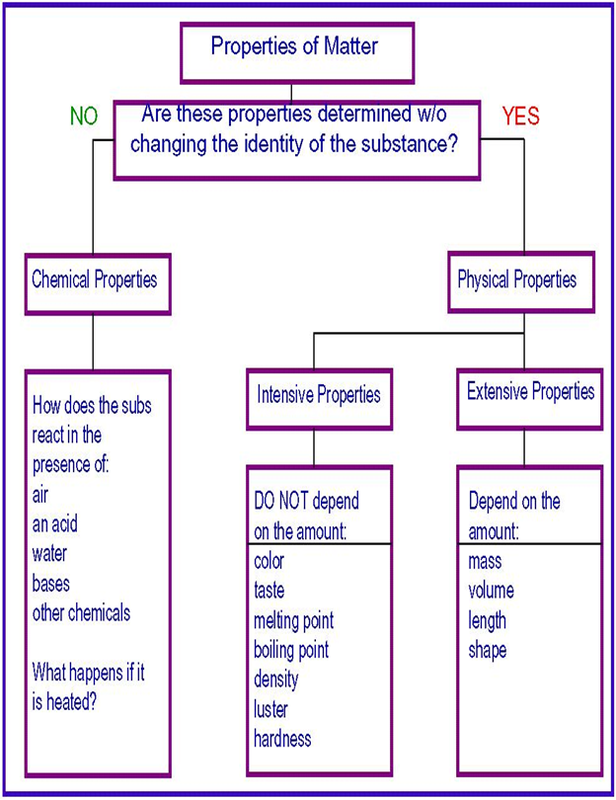
Properties of Matter
Matter has its own unique properties, this could be its chemical property, questions often asked with regards to its chemical property are what is it composed of? How does it react with other substance? Is it an acid or a base? This are the question usually asked when chemical property is talked about. When it comes to Physical property, it is divided into 2 parts, its extensive and intensive property. An intensive property is a bulk property, meaning that it is a physical property of a system that does not depend on the system size or the amount of material in the system. Examples of intensive properties are the temperature and the hardness of an object. No matter how small a diamond is cut, it maintains its intrinsic hardness. While in extensive property, the property is proportional to the amount of material in the system. For example, both the mass and the volume of a diamond are directly proportional to the amount that is left after cutting it from the raw mineral. Mass and volume are extensive properties, but hardness is intensive.
By Jason Angeles
Matter
Matter is anything that occupies space and has mass. Basically, there are Three phases of matter:
Solid - their molecules are more compact and have strong inter-molecular attraction and have less kinetic energy meaning there movement is restricted and can only vibrate, like rocks, metal and ice.
Liquid - liquids have indefinite shape, their molecules are less compact meaning that they can move more freely than solids, and have low kinetic energy. Examples are water, coca cola, and alcohol.
Gas – Gas is the 3rd phase of matter. Their molecules are scattered, meaning they can move more freely and have high kinetic energy. Examples are Oxygen, Carbon dioxide, and helium.
Properties of Matter
Matter has its own unique properties, this could be its chemical property, questions often asked with regards to its chemical property are what is it composed of? How does it react with other substance? Is it an acid or a base? This are the question usually asked when chemical property is talked about. When it comes to Physical property, it is divided into 2 parts, its extensive and intensive property. An intensive property is a bulk property, meaning that it is a physical property of a system that does not depend on the system size or the amount of material in the system. Examples of intensive properties are the temperature and the hardness of an object. No matter how small a diamond is cut, it maintains its intrinsic hardness. While in extensive property, the property is proportional to the amount of material in the system. For example, both the mass and the volume of a diamond are directly proportional to the amount that is left after cutting it from the raw mineral. Mass and volume are extensive properties, but hardness is intensive.